Run Date
Indicates the date and time (Central Time) the report was generated.
Tables
Tables 1, 2, and 3 are summary tables that display a count of queries by CPI time periods, programs, and studies, respectively. Table 4 is a detailed table that provides a list of unresolved queries. Centers can filter Table 4 to locate a specific query.
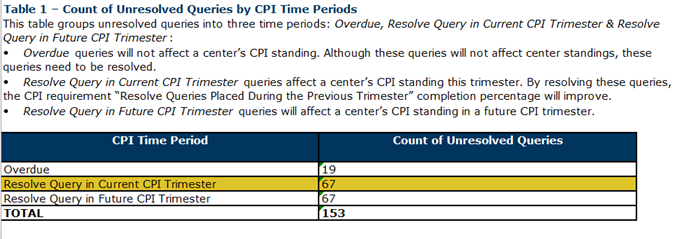
Table 1
Table 1 indicates the count of unresolved queries by CPI Time Periods.
- Overdue: Queries that are overdue (not counted in CPI, but still need to be resolved)
- Current CPI Trimester: Queries that need to be resolved for current CPI trimester (queries placed in last trimester)
- Future CPI Trimester: Queries that will need to be resolved in a future CPI trimester
Table 2
Table 2 indicates the count of unresolved queries by CIBMTR Program.
- CTA: Unresolved queries placed for the consecutive audit program (CTA)
- CVDR: Unresolved queries placed for the center volumes data report (CVDR)
- TCSA: Unresolved queries placed for the transplant center specific analysis (TCSA)
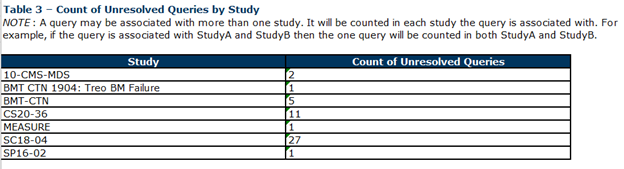
Table 3
Table 3 indicates unresolved queries by study.
Table 4
Table 4 indicates all unresolved queries and detailed information. Centers can filter this table for specific information. This table includes specifics on the following fields:
| Field Name | Description |
| Type of Query | - Form - Consent tool record - CRID Assignment Tool |
| CCN | CIBMTR Center Number (5-digits) |
| CRID | Recipient’s CIBMTR Research ID |
| Form | Form number |
| Visit | Follow-up time point |
| Event Date | Event or infusion date |
| Sequence Number | Unique form identifier |
| Consent ID | Unique consent tool identifier |
| Section | Section name in form |
| Instance | Instance the queried question exists in |
| Question | Question number |
| Question Text | Question text |
| Date Query Placed | Date query was placed |
| Date Query Last Updated | Most recent date the query was interacted with |
| CPI Time Period* | - Legacy query (international centers only) - Overdue - Resolve query in current CPI trimester - Resolve query in future CPI trimester |
| CPI Trimester – to be Resolved | CPI trimester the query needs to be resolved in |
| Program | - CTA - CVDR (U.S. centers only) - TCSA |
| Study | Study number |
| Type of Infusion | - HCT - CT - Genetically-modified HCT - Cellular Therapy (GM CT) - Cellular Therapy (non-GM CT) - Gene Therapy (GT) - Auto Boost |
| Donor Type | - AUTO (Autologous) - ALLO_U (Allogeneic Unrelated) - ALLO_R (Allogeneic Related) - ALLO (Allogeneic) - UNK (Unknown) |
| Institution Specific Subject ID | Recipient’s institution specific subject ID |
| Patient Birth Year | Recipient’s year of birth |
| Disease | Recipient’s primary disease |
| Age of Query (Months) | Number of months since the query was placed |
Last modified:
Mar 03, 2025