What is TCSA?
Transplant Center Specific Analysis (TCSA), also referred to as Center Specific Analysis (CSA) or Center Outcomes Analysis, is used to predict one-year survival of first allogeneic transplants, based on data reported to CIBMTR. This program is mandatory for US centers performing allogeneic transplants. Reporting center-specific survival rates is a requirement of the TRANSPLANT Act of 2021, previously the Stem Cell Therapeutic and Research Act of 2005 (reauthorized in 2010 and 2015), and prior to that, the 1990 Transplant Amendments Act.
The analysis is dependent upon the accuracy and completeness of reporting from centers. Since centers vary considerably in the risk level of cases treated, a statistical model was developed to adjust for several risk factors known or suspected to influence transplant outcomes.
This model is used to predict an “expected” 1-year (365+ days) survival post-first allogeneic transplant for a center, given the types of patients and diseases treated. The actual survival observed at a center is then compared against the “expected” survival. The center will receive one of the following scores: Below expected, Meets expected, or Exceeds expected survival.
Purpose of TCSA
This robust analysis provides an equitable, balanced, and scientific performance measurement tool that can be used by the transplant community to define and improve quality.
The results of this analysis are considered in FACT Accreditation and insurance companies may determine coverage based upon a center’s score.
TCSA Timeline
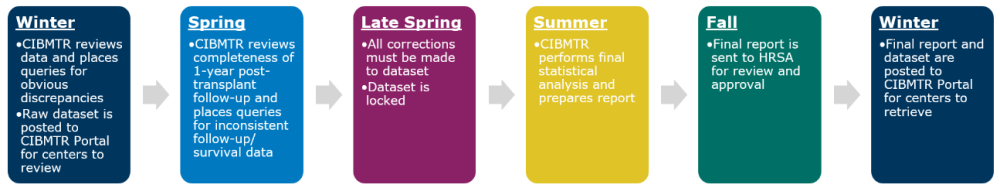
The analysis begins in December with CIBMTR gathering an initial dataset. The dataset includes first related or unrelated donor transplants performed in a three-year time interval.
CIBMTR reviews the initial dataset for incorrect or missing information and places queries in FormsNet3. Raw datasets are then provided for centers to be reviewed in CIBMTR Portal (Portal > DataOps Dashboard> TCSA). The datasets include all allogeneic HCTs performed within the three-year interval, allowing centers to identify patterns and outliers, and to validate HCT history.
These datasets are split into multiple files to make the disease data easier to review. These files include several more variables than the Center Volumes Data Report (CVDR). Centers should make any necessary corrections in FormsNet3 by as soon as practical. Monitor communications for specific deadlines. See Center-Specific Survival Analysis for more information on methodology and FAQs.
In March, CIBMTR begins reviewing completeness of the 1-year follow-up. Queries will be placed for inconsistent follow-up/survival data. Cases must have follow-up reported through one-year post-transplant or report of patient death.
Once the dataset is locked, CIBMTR performs a statistical analysis to prepare the final report sent to Health Resources and Service Administration (HRSA) at the end of summer. After HRSA has granted their approval, the final report is posted to the HRSA public website and CIBMTR Portal. The report is usually available to centers in December. When it is posted on the CIBMTR Portal, the schedule for the following year is also posted.
The timeline below displays a further breakdown of these timeframes.