Each year CIBMTR performs a consecutive transplant audit (CTA) for all active centers to ensure the epidemiological integrity of the CIBMTR outcomes registry and to provide the US government with an accurate assessment of transplant activity. The audit ensures all hematopoietic cell transplantation (HCT) infusions performed in the prior calendar year (January – December), at each active center, have been reported to CIBMTR.
- For example, to be compliant with the CTA process in 2023, centers must include all HCT infusions performed from calendar year January 1, 2022 – December 31, 2022.
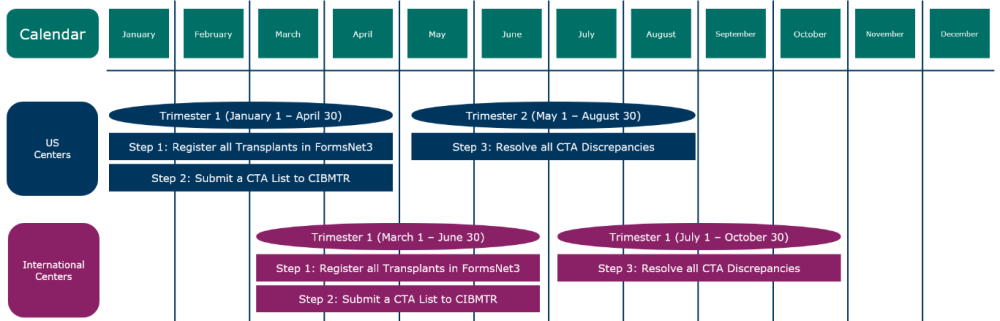
The CTA process requires centers report infusions in FormsNet3, then submit a CTA List to CIBMTR, and lastly, resolve any discrepancies to comply with the audit. Each of these steps aligns with the Continuous Process Improvement (CPI) trimesters. See the CTA timeline diagram below for details.
Centers must meet the CTA requirements to remain in “Good Standing” for CPI. These requirements are not subject to any exemption.
CTA Timeline
![]()
CTA Steps
- Step 1: Report Infusions in FormsNet3
- Step 2: Submit CTA List to CIBMTR
- Step 3: CTA Discrepancy Process