Recipient consent for the CIBMTR research database (ClinicalTrials.gov Identifier: NCT01166009) is collected in the Consent Tool within FormsNet3. The Consent Tool allows recipient consent status and contact information to be collected as soon as possible after the CRID is created. It also enables the CIBMTR Survey Research Group (SRG) to approach patients prior to their infusion to conduct patient-reported outcomes (PRO) surveys.
Figure 1. Disabled Edit Form Icon
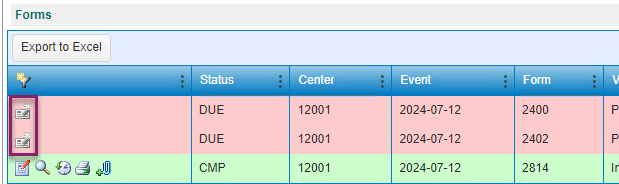
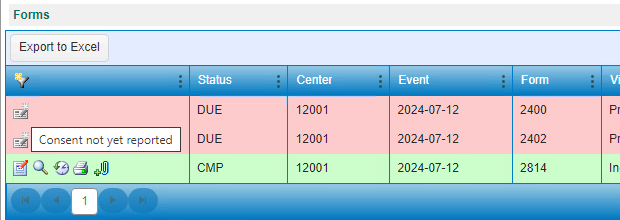
Figure 2. Hovered Text, Consent Not Yet Reported
Links to Sections of the Consent Tool Instructions:
Navigation to the Consent Tool
Consent Tool Grids
Adding and Updating Consent
Q1-10: Consent Information
Last modified:
Jul 26, 2024

